Artificial Intelligence
Methicillin-Resistant Staphylococcus aureus Infection Pipeline Clinical Trials Featuring 25+ companies| DelveInsight
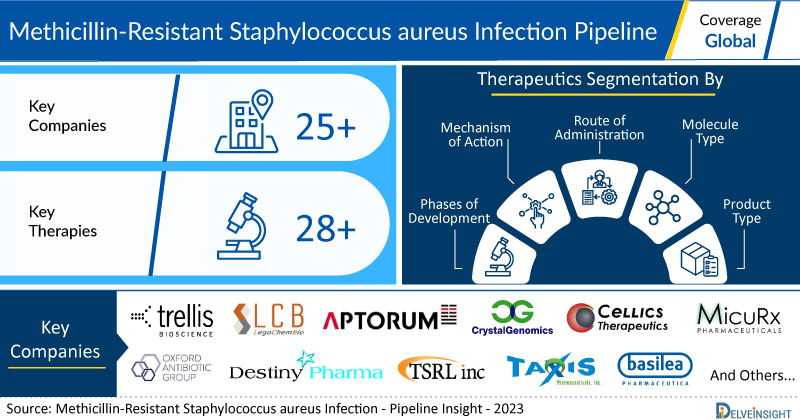
New York, USA, Jan. 25, 2023 (GLOBE NEWSWIRE) — Methicillin-Resistant Staphylococcus aureus Infection Pipeline Clinical Trials Featuring 25+ companies| DelveInsight
Methicillin-resistant Staphylococcus aureus (MRSA) is an infection caused by antibiotic-resistant strains of the pathogen Staphylococcus aureus. Rising incidence of Methicillin-resistant Staphylococcus aureus (MRSA) infections is creating health concerns among the population. Manufacturers are focusing on product development and launches for the treatment of Methicillin-resistant Staphylococcus aureus (MRSA).
DelveInsight’s ‘Methicillin-Resistant Staphylococcus aureus Infection Pipeline Insight 2023‘ report provides comprehensive global coverage of available, marketed, and pipeline methicillin-resistant Staphylococcus aureus infection therapies in various stages of clinical development. Major pharmaceutical companies are working to advance the pipeline space and future growth potential of the methicillin-resistant Staphylococcus aureus infection pipeline domain.
Key Takeaways from the Methicillin-Resistant Staphylococcus aureus Infection Pipeline Report
- DelveInsight’s methicillin-resistant Staphylococcus aureus infection pipeline report depicts a robust space with 25+ companies and 28+ pipeline drugs for methicillin-resistant Staphylococcus aureus infection treatment.
- Key methicillin-resistant Staphylococcus aureus infection companies include Trellis Bioscience, LegoChem Biosciences, Aptorum Group, ContraFect, Crystalgenomics, Cellics Therapeutics, MicuRx, Oxford Antibiotic Group, Destiny Pharma, TSRL, Inc., TAXIS Pharmaceuticals, Basilea Pharmaceutica, Helperby Therapeutics, Akagera Medicines, Histogen, Alphamab Co. Ltd, Biocidium Pharmaceuticals, and others are evaluating novel methicillin-resistant Staphylococcus aureus infection drugs candidate to improve the treatment landscape.
- Promising methicillin-resistant Staphylococcus aureus infection pipeline therapies in various stages of development include TRL1068, Delpazolid, ALS 4, Teicoplanin inhalation, CG 400549, SAAP 148, Exebacase, Marinus, CTI 005, Iclaprim, MRX 4, MRX 7, XF-73, TXA709, ARV-1801, ACX-375C, ABX-605, IBT-V02, BCM-0184, Contezolid, TetramOX, Ceftobiprole, HY-004B8b, HY-005B8a, CG-549, Emricasan, KN043, and several others.
- Ceftobiprole medocaril, the prodrug of the active moiety ceftobiprole, is a cephalosporin antibiotic for intravenous administration, with rapid bactericidal activity against a wide range of Gram-positive and Gram-negative bacteria. This includes methicillin-susceptible and resistant Staphylococcus aureus (MSSA, MRSA) and susceptible Pseudomonas spp. In October 2022, Basilea Pharmaceutica announced the results from Phase III ERADICATE study, evaluating ceftobiprole in the treatment of adult patients with bacterial bloodstream infections caused by Staphylococcus aureus. Results for the primary efficacy outcomes were consistent in key subgroups including in patients with MSSA or MRSA bloodstream infections at baseline, and in various categories of underlying conditions such as skin and skin structure infections, abdominal abscesses, chronic dialysis, septic arthritis, osteomyelitis, definite right-sided infective endocarditis and in patients with persistent SAB. Basilea also planned to submit a New Drug Application (NDA) for ceftobiprole to the US Food and Drug Administration (FDA) around year-end 2022.
- Exebacase (CF-301) being developed by ContraFect Corporation is a lysin with potent activity against Staphylococcus aureus (“Staph aureus”). Exebacase has the potential to be the first-in-class of a new treatment for patients with Staph aureus bacteremia. The FDA has granted Breakthrough Therapy designation to exebacase for the treatment of MRSA bloodstream infections (bacteremia), including right-sided endocarditis, when used in addition to SOC antistaphylococcal antibiotics in adult patients, based on the final data from the Phase II superiority trial of exebacase.
- In July 2022, Destiny Pharma plc announced that it had received an award from the Cystic Fibrosis Foundation. The research project will establish the potential of the Company’s proprietary XF-73 drug as a novel treatment for cystic fibrosis patients infected with methicillin-resistant Staphylococcus aureus (MRSA).
- CTI-005 is for the treatment of Methicillin Resistance Staphylococcus Aureus (MRSA) Pneumonia and has been granted IND allowance from the US FDA. The potent RBC-NS has the membrane lipids and receptors that a normal red blood cell has, however, the high surface-volume ratio of the RBC-NS allows it to act as a decoy. This characteristic allows the RBC-NS to bind and neutralize toxins, auto-antibodies, and bacterial infections. The pore-forming toxins emitted by MRSA can be effectively absorbed and neutralized by RBC-NS.
Request a sample and discover the recent advances in methicillin-resistant Staphylococcus aureus infection treatment drugs @ Methicillin-Resistant Staphylococcus aureus Infection Pipeline Report
The methicillin-resistant Staphylococcus aureus infection pipeline report provides detailed profiles of pipeline assets, a comparative analysis of clinical and non-clinical stage methicillin-resistant Staphylococcus aureus infection products, inactive and dormant assets, a comprehensive assessment of driving and restraining factors, and an assessment of opportunities and risks in the methicillin-resistant Staphylococcus aureus infection pipeline landscape.
Methicillin-Resistant Staphylococcus aureus Infection Overview
Methicillin-resistant Staphylococcus aureus (MRSA) infections are caused by bacteria that have attained resistance from several antibiotics that treat diseases caused by Staphylococcus bacteria. The common patients of Methicillin-resistant Staphylococcus aureus (MRSA) infections are patients that have been in hospitals or healthcare settings for a long duration where the bacteria is found commonly. MRSA infections are known as healthcare-associated MRSA (HA-MRSA) if infected in hospitals. Such infections can spread to healthcare workers through invasive procedures, which are further transmitted to several patients by touching and operating with bare hands.
Other MRSA infections occur in a considerable population of healthy people. People associated with community-associated MRSA (CA-MRSA) show symptoms appearing as painful skin boils. It further spreads through physical contact between individuals. The population at higher risk includes people surrounded by a significant number of people and childcare workers. MRSA infections might look like red bumps, pimples, or spider bites, accompanied by pus and fever.

Find out more about methicillin-resistant Staphylococcus aureus infection treatment drugs @ Drugs for Methicillin-Resistant Staphylococcus aureus Infection Treatment
A snapshot of the Methicillin-Resistant Staphylococcus aureus Infection pipeline drugs mentioned in the report:
| Drug | Company | Phase | MoA | RoA |
| Ceftobiprole | Basilea Pharmaceutica | Phase III | Cell wall inhibitors; Peptidyltransferase inhibitors | Intravenous |
| CG 549 | CrystalGenomics
|
Phase II | Enoyl-ACP reductase inhibitors; Fatty acid synthase expression inhibitors; Fatty acid synthetase complex inhibitors | Oral |
| BCM-0184 | Biocidium Pharmaceuticals | Phase I | NA | Oral/Topical |
| TRL1068 | Trellis Bioscience | Phase I | Bacterial biofilm inhibitors | Intravenous |
| ALS 4 | Aptorum Group | Phase I | Bacterial virulence inhibitors | Oral |
| EVX-B1 | Evaxion Biotech | Preclinical | Immunostimulants | NA |
| Emricasan | Histogen | Preclinical | Apoptosis inhibitors; Caspase inhibitors | NA |
| TarO inhibitor | Prokaryotics | Discovery | Cell membrane inhibitors; Cell wall inhibitors | NA |
Learn more about the emerging methicillin-resistant Staphylococcus aureus infection pipeline therapies @ Methicillin-Resistant Staphylococcus aureus Infection Clinical Trials
Methicillin-Resistant Staphylococcus aureus Infection Therapeutics Assessment
The methicillin-resistant Staphylococcus aureus infection pipeline report proffers an integral view of methicillin-resistant Staphylococcus aureus infection emerging novel therapies segmented by stage, product type, molecule type, mechanism of action, and route of administration.
Scope of the Methicillin-Resistant Staphylococcus aureus Infection Pipeline Report
- Coverage: Global
- Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
- Therapeutic Assessment By Clinical Stages: Pre-clinical, Discovery, Discontinued, Inactive, Phase I, Phase II, Phase III
- Therapeutics Assessment By Route of Administration: Oral, Parenteral, Intravenous, Subcutaneous, Topical.
- Therapeutics Assessment By Molecule Type: Monoclonal Antibody, Peptides, Polymer, Small molecule, Gene therapy
- Therapeutics Assessment By Mechanism of Action: Cell wall inhibitors; Peptidyltransferase inhibitors, Enoyl-ACP reductase inhibitors; Fatty acid synthase expression inhibitors; Fatty acid synthetase complex inhibitors, Bacterial biofilm inhibitors, Bacterial virulence inhibitors, Immunostimulants, Apoptosis inhibitors; Caspase inhibitors, Cell membrane inhibitors; Cell wall inhibitors
- Key Methicillin-Resistant Staphylococcus aureus Infection Companies: Trellis Bioscience, LegoChem Biosciences, Aptorum Group, ContraFect, Crystalgenomics, Cellics Therapeutics, MicuRx, Oxford Antibiotic Group, Destiny Pharma, TSRL, Inc., TAXIS Pharmaceuticals, Basilea Pharmaceutica, Helperby Therapeutics, Akagera Medicines, Histogen, Alphamab Co. Ltd, Biocidium Pharmaceuticals, and others.
- Key Methicillin-Resistant Staphylococcus aureus Infection Pipeline Therapies: TRL1068, Delpazolid, ALS 4, Teicoplanin inhalation, CG 400549, SAAP 148, Exebacase, Marinus, CTI 005, Iclaprim, MRX 4, MRX 7, XF-73, TXA709, ARV-1801, ACX-375C, ABX-605, IBT-V02, BCM-0184, Contezolid, TetramOX, Ceftobiprole, HY-004B8b, HY-005B8a, CG-549, Emricasan, KN043, and others.
Dive deep into rich insights for new drugs for methicillin-resistant Staphylococcus aureus infection treatment; visit @ Methicillin-Resistant Staphylococcus aureus Infection Drugs
Table of Contents
| 1. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline Report Introduction |
| 2. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline Report Executive Summary |
| 3. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline: Overview |
| 4. | Analytical Perspective In-depth Commercial Assessment |
| 5. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline Therapeutics |
| 6. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline: Late Stage Products (Pre-registration) |
| 7. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline: Late Stage Products (Phase III) |
| 7.1 | Ceftobiprole: Basilea Pharmaceutica |
| 8. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline: Mid Stage Products (Phase II) |
| 8.1 | CG549: CrystalGenomics |
| 9. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline: Early Stage Products (Phase I) |
| 9.1 | TRL1068: Trellis Bioscience |
| 10. | Methicillin-Resistant Staphylococcus aureus Infection Pipeline Therapeutics Assessment |
| 11. | Inactive Products in the Methicillin-Resistant Staphylococcus aureus Infection Pipeline |
| 12. | Company-University Collaborations (Licensing/Partnering) Analysis |
| 13. | Key Companies |
| 14. | Key Products in the Methicillin-Resistant Staphylococcus aureus Infection Pipeline |
| 15. | Unmet Needs |
| 16. | Market Drivers and Barriers |
| 17. | Future Perspectives and Conclusion |
| 18. | Analyst Views |
| 19. | Appendix |
For further information on the methicillin-resistant Staphylococcus aureus infection pipeline therapeutics, reach out @ Methicillin-Resistant Staphylococcus aureus Infection Treatment Drugs
Related Reports
Methicillin-Resistant Staphylococcus aureus Epidemiology Forecast
Methicillin-Resistant Staphylococcus aureus Epidemiology Forecast – 2032 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, and the methicillin-resistant Staphylococcus aureus epidemiology trends.
Methicillin-Resistant Staphylococcus aureus Infection Market
Methicillin-Resistant Staphylococcus aureus Market Insights, Epidemiology, and Market Forecast – 2032 report delivers an in-depth understanding of the market trends, market drivers, market barriers, and key methicillin-resistant Staphylococcus aureus infection companies, including Trellis Bioscience, LegoChem Biosciences, Aptorum Group, Allergan, iNtRON Biotechnology, Neupharma, ImmuPharma, Lytix Biopharma, CrystalGenomics, AIMM Therapeutics, among others.
Staphylococcus aureus Bacteremia Pipeline
Staphylococcus aureus Bacteremia Pipeline Insight – 2023 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key Staphylococcus aureus Bacteremia companies, including ContraFect, Armata Pharmaceuticals, iNtRON Biotechnology, Durata Therapeutics, Cipher Pharmaceuticals, XBiotech, Basilea Pharmaceutica, Genentech, among others.
Hospital Acquired Methicillin-Resistant Staphylococcus aureus Infections Pipeline
Hospital Acquired Methicillin-Resistant Staphylococcus aureus Infections Pipeline Insight – 2023 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key Hospital Acquired Methicillin-Resistant Staphylococcus aureus Infections companies, including Trellis Bioscience, LegoChem Biosciences, Aptorum Group, Allergan, iNtRON Biotechnology, Neupharma, ImmuPharma, Lytix Biopharma, CrystalGenomics, AIMM Therapeutics, among others.
Other Trending Reports
Treatment Resistant Depression Market | Advanced Renal Cell Carcinoma Market | Brain Cancer Market | Centronuclear Myopathy Market | Head And Neck Squamous Cell Carcinoma Market | Acquired Immunodeficiency Syndrome Market | Acute Intermittent Porphyria Market | Neurovascular Devices Market | Defibrillators Market | Ventricular Hypertrophy Market | Urolithiasis Market | Alopecia Areata Market | Autonomic Dysfunction Market | Acute Ischemic Stroke Ais Market | Adenoid Cystic Carcinoma Market | Aspergillosis Market | Biliary Atresia Market | Biliary Tumor Market | Chronic Inducible Urticaria Market | Chronic Insomnia Market | Critical Limb Ischemia Market | Endometriosis Pain Market | Generalized Anxiety Disorder Gad Market | Hallux Valgus Market | Hemophilia B Market | Immunologic Deficiency Syndrome Market | Neuroblastoma Market | Neuromodulation Devices Market | Neurovascular Thrombectomy Devices Market | Osteosarcoma Market | Pemphigus Vulgaris Market | Progressive Familial Intrahepatic Cholestasis Market | Pruritus Market | Radiation Toxicity Market | Pulmonary Hypertension Associated With Interstitial Lung Disease Market | Cluster Headaches Market | Foot And Ankle Devices Market | Intravenous Immunoglobulin Market | Bile Duct Neoplasm Market | Blood Glucose Monitoring Systems Market | Rett Syndrome Market | Tissue Heart Valves Market | Cardiac Biomarkers Testing Devices Market | Subscription Healthcare | Hepatorenal Syndrome Market | Central Venous Catheters Market | Continuous Renal Replacement Therapy Machines Market | Nonalcoholic Steatohepatitis Market | Necrotizing Enterocolitis Market | Cardiac Amyloidosis Market | Artificial Iris Market | Aortic Aneurysm Stent Grafts Market | Polypoidal Choroidal Vasculopathy Market | Adrenal Crisis Market | Hearing Implants Market | Image Guided Surgery Devices Market | Angioedema Market | Bladder Cancer Market
Related Healthcare Services
Healthcare Competitive Intelligence Services
Healthcare Asset Prioritization Services
About DelveInsight
DelveInsight is a leading Business Consultant, and Market Research firm focused exclusively on life sciences.
Connect with us at LinkedIn

Artificial Intelligence
Data Center Chip Market Size was Valued at USD 11.7 Billion in 2022 and is Expected to Reach USD 45.3 Billion by 2032 at a CAGR of 14.6% | Valuates Reports
BANGALORE, India, July 26, 2024 /PRNewswire/ — Data Center Chip Market By Chip Type (GPU, ASIC, FPGA, CPU, Others), By Data Center Size (Small and Medium Size, Large Size), By Industry Verticals (BFSI, Manufacturing, Government, IT and Telecom, Retail, Transportation, Energy and Utilities, Others): Global Opportunity Analysis and Industry Forecast, 2023-2032.
The Data Center Chip Market was valued at USD 11.7 Billion in 2022, and is estimated to reach USD 45.3 Billion by 2032, growing at a CAGR of 14.6% from 2023 to 2032.
Get Free Sample @ https://reports.valuates.com/request/sample/ALLI-Auto-2B326/Data_Center_Chip_Market
Major Factors Driving the Growth of Data Center Chip Market
Because of the growing need for data processing and storage solutions brought about by the quick development of cloud computing, artificial intelligence, and big data analytics, the data center chip market is expanding significantly. High-performance chips are necessary for data centers to process massive volumes of data quickly and efficiently. As a result, advances in chip technology, including CPUs, GPUs, and specialist AI processors, have been made. The need for more resilient and scalable data center infrastructure is fueled in part by the expansion of digital services and Internet of Things (IoT) devices. The market is expanding due to key areas including Asia-Pacific, with its investments in technology and fast digital transformation, and North America, with its top tech businesses and vast data center networks.
View Full Report @ https://reports.valuates.com/market-reports/ALLI-Auto-2B326/data-center-chip
TRENDS INFLUENCING THE GROWTH OF THE DATA CENTER CHIP MARKET:
In data centers, Graphics Processing Units (GPUs) are essential for speeding up computing operations and data processing. They are perfect for managing workloads related to artificial intelligence (AI), machine learning, and large-scale data analytics because of their parallel processing capabilities. The need for GPUs in data centers is growing as these technologies become increasingly essential to corporate operations. Businesses are purchasing GPUs in order to increase the effectiveness of their data processing, lower latency, and boost overall performance. The need for data center chips is being driven by the increasing reliance on GPUs for sophisticated computing activities, which is considerably contributing to the market’s rise. This need is further increased by the growing use of AI and machine learning in a variety of sectors, which puts GPUs at the forefront of the data center semiconductor industry.
Compared to general-purpose chips, Application Specific Integrated Circuits (ASICs) provide better performance and efficiency since they are designed specifically for a given application. ASICs are extensively utilized in data centers for specific tasks including networking, data compression, and encryption. ASICs are becoming more and more common as a result of the growth of cloud computing, big data analytics, and blockchain technology, which has increased demand for high-performance, energy-efficient processors. Their capacity to provide tailored performance for certain applications aids data centers in better workload management, power conservation, and operating expense reduction. The market is expanding as a result of the increased preference for ASICs in data centers, which is fueling the need for specialized data center chips.
Large data centers are important users of data center chips; they are run by well-known IT firms and cloud service providers. To manage enormous volumes of data and provide a wide range of services, these facilities need a great deal of processing power and sophisticated computing skills. High-performance data center chips are becoming more and more necessary as a result of the growth of massive data centers and the rising demand for online streaming, cloud services, and digital transactions. These chips are necessary to ensure effective data management, processing, and storage, which helps big data centers fulfill the increasing expectations of its clientele. Large data center proliferation is anticipated to considerably boost the data center chip industry as the digital economy continues to grow.
Data centers are becoming more and more important to the Banking, Financial Services, and Insurance (BFSI) industry as a means of safely and effectively managing high transaction volumes, consumer data, and financial records. The need for sophisticated data center processors is being driven by the sector’s requirement for real-time data processing, high-performance computing, and strong security measures. BFSI organizations may improve their operational efficiency, guarantee data integrity, and deliver superior client services by utilizing data centers fitted with robust chips. The BFSI sector’s need for data center chips is being driven by the increasing use of online banking, digital banking, and financial analytics tools, all of which increase the requirement for sophisticated data center infrastructure.
The market for data center chips is significantly influenced by the cloud computing industry’s explosive growth. There is a growing need for scalable, effective, and high-performance data center infrastructure as more companies move their operations to the cloud. In order to handle enormous volumes of data, facilitate virtualization, and guarantee flawless service delivery, cloud service providers need sophisticated data center chips. Sturdy data center chips are becoming more and more necessary as cloud-based solutions become more and more popular. Benefits like cost savings, flexibility, and scalability are driving this trend. In places like North America and Europe, where cloud adoption rates are high and data center chip demand is rising rapidly, this tendency is especially significant.
Buy Now @ https://reports.valuates.com/api/directpaytoken?rcode=ALLI-Auto-2B326&lic=single-user
DATA CENTER CHIP MARKET SHARE
In 2022, North America gained a sizable portion of the market.
In 2022, the GPU made up the largest portion of the market share.
Throughout the projection period, large data centers are expected to gain a significant portion.
The BFSI market is anticipated to be one of the most profitable markets.
Purchase Regional Report @ https://reports.valuates.com/request/regional/ALLI-Auto-2B326/Data_Center_Chip_Market
Key Companies:
Advanced Micro Devices IncTaiwan Semiconductor Manufacturing Company LimitedBroadcomHuawei Technologies Co LtdIntel CorporationNVidia CorporationSamsung Electronics Co LtdQualcomm Technologies IncGlobalFoundriesARM LIMITED (SOFTBANK GROUP CORP.)Purchase Chapters @ https://reports.valuates.com/request/chaptercost/ALLI-Auto-2B326/Data_Center_Chip_Market
SUBSCRIPTION
We have introduced a tailor-made subscription for our customers. Please leave a note in the Comment Section to know about our subscription plans.
DISCOVER MORE INSIGHTS: EXPLORE SIMILAR REPORTS!
– The global modular data center market size was valued at USD 14,952 Million in 2019 and is projected to reach USD 59,971 Million by 2027, registering a CAGR of 18.7% from 2020 to 2027.
– Data Centre Market was estimated to be worth USD 137500 Million in 2023 and is forecast to a readjusted size of USD 412740 Million by 2030 with a CAGR of 16.8% during the forecast period 2024-2030.
– Data Center PCIe Chip market was valued at USD 194.9 Million in 2023 and is anticipated to reach USD 377.4 Million by 2030, witnessing a CAGR of 10.2% during the forecast period 2024-2030.
– Data Center Accelerator Market
– Data Center Networking Market
– According to a new report published by , titled, “Big Data Analytics in Semiconductor & Electronics Market,” The big data analytics in semiconductor & electronics market was valued at D18.7 billion in 2021, and is estimated to reach D47.2 billion by 2031, growing at a CAGR of 9.9% from 2022 to 2031.
– IoT market was valued at USD 34250 Million in 2023 and is anticipated to reach USD 74630 Million by 2030, witnessing a CAGR of 11.6% during the forecast period 2024-2030.
– Data Center AI Accelerator Chip Market
– Electro-absorption Modulated Laser Chip Market
– According to a new report published by , titled, “Data Processing Unit Market”, the data processing unit market was valued at D553.96 Million in 2021, and is estimated to reach D5.5 billion by 2031, growing at a CAGR of 26.9% from 2022 to 2031.
– Optical Chip for Data Center Market
– EML Chip Market
– Optical Communication Chip Market revenue was USD 3102.7 Million in 2022 and is forecast to a readjusted size of USD 7251.5 Million by 2029 with a CAGR of 12.9% during the forecast period (2023-2029).
– SiC Power Chip Market
– Silicon Carbide Chip Market
– High Speed Optical Communication Chip Market
– 56G eml Optical Chip Market
– FPGA Master Control Chip Market
– Single-port Industrial Grade Ethernet PHY Chip Market
– AWG Chip Market
– Discrete Graphics Chip Market
– LNOI Optoelectronic Chip Market
– Smartphone Chip Market
– Smartphone TFT-LCD Display Driver Chip Market
– Smart Phone Baseband Chip Market
DISCOVER OUR VISION: VISIT ABOUT US!
Valuates offers in-depth market insights into various industries. Our extensive report repository is constantly updated to meet your changing industry analysis needs.
Our team of market analysts can help you select the best report covering your industry. We understand your niche region-specific requirements and that’s why we offer customization of reports. With our customization in place, you can request for any particular information from a report that meets your market analysis needs.
To achieve a consistent view of the market, data is gathered from various primary and secondary sources, at each step, data triangulation methodologies are applied to reduce deviance and find a consistent view of the market. Each sample we share contains a detailed research methodology employed to generate the report. Please also reach our sales team to get the complete list of our data sources.
YOUR FEEDBACK MATTERS: REACH OUT TO US!Valuates [email protected] U.S. Toll-Free Call 1-(315)-215-3225WhatsApp: +91-9945648335Website: https://reports.valuates.comBlog: https://valuatestrends.blogspot.com/ Pinterest: https://in.pinterest.com/valuatesreports/ Twitter: https://twitter.com/valuatesreports Facebook: https://www.facebook.com/valuatesreports/ YouTube: https://www.youtube.com/@valuatesreports6753 https://www.facebook.com/valuateskorean https://www.facebook.com/valuatesspanish https://www.facebook.com/valuatesjapanese https://valuatesreportspanish.blogspot.com/ https://valuateskorean.blogspot.com/ https://valuatesgerman.blogspot.com/ https://valuatesreportjapanese.blogspot.com/
Logo: https://mma.prnewswire.com/media/1082232/Valuates_Reports_Logo.jpg
View original content:https://www.prnewswire.co.uk/news-releases/data-center-chip-market-size-was-valued-at-usd-11-7-billion-in-2022-and-is-expected-to-reach-usd-45-3-billion-by-2032-at-a-cagr-of-14-6–valuates-reports-302207745.html
Artificial Intelligence
Industry 4.0 Market to Surpass USD 513.89 Billion by 2031 with Automation Surge | SkyQuest Technology
WESTFORD, Mass., July 26, 2024 /PRNewswire/ — According to SkyQuest, the global Industry 4.0 Market size was valued at USD 133.05 billion in 2022 and is poised to grow from USD 154.6 billion in 2023 to USD 513.89 billion by 2031, growing at a CAGR of 16.2% during the forecast period (2024-2031).
Industry 4.0 or the fourth industrial revolution emphasizes the use of automation and interconnectivity. Employment of advanced technologies such as artificial intelligence, machine learning, robotics, and connected devices to improve the productivity and efficiency of industries. Rapid digitization and advancements in technology are forecasted to bolster the Industry 4.0 market growth over the coming years. The global Industry 4.0 market is segmented into technology, industry vertical, and region.
Download a detailed overview:
https://www.skyquestt.com/sample-request/industry-4-0-market
Industry 4.0 Market Overview:
Report Coverage
Details
Market Revenue in 2023
$ 154.6 billion
Estimated Value by 2031
$ 513.89 billion
Growth Rate
Poised to grow at a CAGR of 16.2%
Forecast Period
2024–2031
Forecast Units
Value (USD Billion)
Report Coverage
Revenue Forecast, Competitive Landscape, Growth Factors, and Trends
Segments Covered
Technology, Industry and Region
Geographies Covered
North America, Europe, Asia Pacific, Latin America, and Middle East and Africa.
Report Highlights
Internet of Things (IoT) technology takes centerstage for Industry 4.0 adoption
Key Market Opportunities
Adoption of smart manufacturing and additive manufacturing practices
Key Market Drivers
Rising demand for automation across all industry verticals
Segments covered in Industry 4.0 Market are as follows:
TechnologyRobots (Traditional Industrial Robots {Articulated robots, Cartesian Robots, Selective Compliance Assembly Robot Arm (SCARA), Cylindrical Robots, Others}, Collaborative Robots), Blockchain in Manufacturing, Industrial Sensors (Level Sensors, Temperature Sensors, Flow Sensors, Position Sensors, Pressure Sensors, Force Sensors, Humidity & Moisture Sensors, Gas Sensors), Industrial 3D Printing, Machine Vision (Camera {Digital Camera, Smart Camera}, Frame Grabbers, Optics, and LED Lighting, Processor and Software), HMI (Offering {Hardware [Basic HMI, Advanced Panel-based HMI, Advanced PC-based HMI, Others], Software [On-premises HMI, Cloud-based HMI], Services}), Configuration ({Embedded HMI, Standalone HMI}, Technology {Motion HMI, Bionic HMI, Tactile HMI, Acoustic HMI}, End-user Industry {Process industries [Oil & Gas, Food & beverages, Pharmaceuticals, Chemicals, Energy & power, Metals & mining, Water & wastewater, Others], Discrete industry [Automotive, Aerospace & defense, Packaging, Medical devices, Semiconductor & electronics, Others]}), AI In Manufacturing (Offering {Hardware [Processor MPU, GPU, FPGA, ASIC, Memory, Network], Software [AI solutions- | On-premises, Cloud |, AI platform- | Machine learning framework, Application program interface |], Services [Deployment & integration, Support & maintenance]}, Technology {Machine learning [Deep learning, Supervised learning, Reinforcement learning, Reinforcement learning, Others], Natural language processing [Context-aware computing, Computer vision]}, Application {Predictive maintenance and machinery inspection, Material movement, Production planning, Field services, Quality control, Cybersecurity, Industrial robots, Reclamation}, Digital Twin {Technology [Internet of Things (IOT), Blockchain, Artificial intelligence & machine learning, Artificial intelligence & machine learning, Big data analytics, 5G], Usage Type [Product digital twin, Process digital twin, System digital twin], Application [Product design & development, Performance monitoring, Predictive maintenance, Inventory management, Business optimization, Others]}, Automated Guided Vehicles (AGV) {Type [Tow vehicles, Unit load carriers, Pallet trucks, Assembly line vehicles, Forklift trucks, Others], Navigation Technology [Laser guidance, Magnetic guidance, Inductive guidance, Optical tape guidance, Vision guidance, Others]}, Machine Condition Monitoring {Monitoring Technique [Vibration monitoring, Embedded systems, Vibration analyzers and meters, Thermography, Oil analysis, Corrosion monitoring, Ultrasound emission, Motor current analysis], Offering [Hardware – Vibration sensors, Accelerometers, Tachometers, Infrared sensors, Spectrometers, Ultrasound detectors, Spectrum analyzers, Corrosion probes], Software [Data integration, Diagnostic reporting, Order tracking analysis, Parameter calculation], Deployment Type [On-premises deployment, Cloud deployment], Monitoring Process [Online condition monitoring, Portable condition monitoring]})IndustryManufacturing, Automotive, Energy, Medical, Semiconductor & Electronics, Food & Beverage, Oil & Gas, Aerospace, Metals & Mining, Chemicals, and OthersRequest Free Customization of this report:
https://www.skyquestt.com/speak-with-analyst/industry-4-0-market
Internet of Things (IoT) Technology to Remain Indispensable for Industry 4.0
Internet of Things (IoT) remains the most crucial technology in global Industry 4.0 market growth owing to its role in interconnectivity and automation across different verticals. Advancements in connectivity technologies and rising use of automation in different industry verticals are also estimated to help this sub-segment gain an impressive market share. Surging demand for predictive maintenance will also boost the adoption of IoT technology in the long run.
Advanced robotic technologies are also slated to gain traction in the Industry 4.0 market. Growing acceptance of robots and high investments in advancements of robotic technologies are also slated to create new opportunities for providers of advanced robotics in the Industry 4.0 market. The low margin of error and the immense scope of automation are key benefits of robotics that help this sub-segment flourish.
Artificial intelligence (AI) will be another popular technology in the Industry 4.0 world going forward. Increasing demand for continuous monitoring, real-time analytics, and predictive maintenance are slated to help the demand for artificial intelligence in the future. The rising use of IoT devices will also boost the demand for cloud computing technology in the long run.
View report summary and Table of Contents (TOC):
https://www.skyquestt.com/report/industry-4-0-market
Manufacturing Vertical to Spearhead Industry 4.0 Market Development
The manufacturing vertical is estimated to be at the forefront when it comes to Industry 4.0 adoption. The surge in use of robotics, advanced technologies, and smart manufacturing practices sets the tone for Industry 4.0 in this industry vertical. High emphasis on improving manufacturing efficiency, reducing downtime, and maximizing profits are all contributing to the high market share of this sub-segment.
The automotive industry is another vertical where Industry 4.0 market players could invest to get good returns. The high adoption of advanced robotics and other smart manufacturing technologies to maximize production allows this sub-segment to become a crucial one for Industry 4.0 providers. The aerospace and defense industry vertical also shows a lot of promise for Industry 4.0 companies going forward. Growing demand for advanced manufacturing techniques and technologies to create complex aerospace components is helping Industry 4.0 market growth via this segment.
The oil & gas industry is also estimated to embrace Industry 4.0 trend with open hands as they try to improve their operations and promote better resource utilization. High demand for predictive maintenance to reduce downtime and the growing adoption of digital oilfield solutions are estimated to bolster Industry 4.0 market development in the long run.
To sum it up, the application scope for Industry 4.0 is endless as automation and digitization pick up pace around the world. High investments in development of IoT and AI technologies will create better opportunities for Industry 4.0 companies in the future. The manufacturing industry will remain the top revenue generating sub-segment and more opportunities for aerospace, automotive, and oil & gas verticals will be seen over the coming years.
Related Report:
Digital Twin Market
Cyber Security Market
Artificial Intelligence (AI) Market
Internet Of Things (IoT) Market
Machine Learning Market
About Us:
SkyQuest is an IP focused Research and Investment Bank and Accelerator of Technology and assets. We provide access to technologies, markets and finance across sectors viz. Life Sciences, CleanTech, AgriTech, NanoTech and Information & Communication Technology.
We work closely with innovators, inventors, innovation seekers, entrepreneurs, companies and investors alike in leveraging external sources of R&D. Moreover, we help them in optimizing the economic potential of their intellectual assets. Our experiences with innovation management and commercialization has expanded our reach across North America, Europe, ASEAN and Asia Pacific.
Contact: Mr. Jagraj SinghSkyQuest Technology1 Apache Way,Westford,Massachusetts 01886USA (+1) 351-333-4748Email: [email protected] Our Website: https://www.skyquestt.com/
Logo : https://mma.prnewswire.com/media/2446095/SkyQuest_Logo.jpg
View original content:https://www.prnewswire.co.uk/news-releases/industry-4-0-market-to-surpass-usd-513-89-billion-by-2031-with-automation-surge–skyquest-technology-302206499.html
Artificial Intelligence
Generative AI Cybersecurity Market worth $40.1 billion by 2030 – Exclusive Report by MarketsandMarkets™
CHICAGO, July 26, 2024 /PRNewswire/ — The Generative AI cybersecurity Market is anticipated to experience substantial expansion, ascending from a value of USD 7.1 billion in 2024 to a substantial worth of USD 40.1 billion by the year 2030, according to a new report by MarketsandMarkets™. This growth trajectory reflects a robust compound annual growth rate (CAGR) of 33.4% over the forecast period.
Browse in-depth TOC on “Generative AI cybersecurity Market”
350 – Tables 60 – Figures450 – Pages
Download PDF Brochure @ https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=164202814
Scope of the Report
Report Metrics
Details
Market size available for years
2019–2030
Base year considered
2023
Forecast period
2024–2030
Forecast units
USD (Million)
Segments Covered
Offering, Generative AI-based Cybersecurity, Cybersecurity for Generative AI, Security Type, End-user, and Region
Geographies covered
North America, Europe, Asia Pacific, Middle East & Africa, and Latin America
Companies covered
Microsoft (US), IBM (US), Google (US), SentinelOne (US), AWS (US), NVIDIA (US), Cisco (US), CrowdStrike (US), Fortinet (US), Zscaler (US), Trend Micro (Japan), Palo Alto Networks (US), BlackBerry (Canada), Darktrace (UK), F5 (US), Okta (US), Sangfor (China), SecurityScorecard (US), Sophos (UK), Broadcom (US), Trellix (US), Veracode (US), LexisNexis (US), Abnormal Security (US), Adversa AI (Israel), Aquasec (US), BigID (US), Checkmarx (US), Cohesity (US), Credo AI (US), Cybereason (US), DeepKeep (Israel), Elastic NV (US), Flashpoint (US), Lakera (US), MOSTLY AI (Austria), Recorded Future (US), Secureframe (US), Skyflow (US), SlashNext (US), Snyk (US), Tenable (US), TrojAI (Canada), VirusTotal (Spain), XenonStack (UAE), and Zerofox (US).
This dramatic surge is being fueled by a number of causes. The primary growth driver is the enhancement of existing cybersecurity tools through generative AI algorithms by improving anomaly detection, automating threat hunting and penetration testing, and providing complex simulations for security testing purposes. These techniques enable various cyber-attack scenarios that can be simulated using the Generative Adversarial Networks (GANs), thus enabling the development of better preparedness and response strategies. On the other hand, it requires special cyber security tools to protect generative AI workloads against unique vulnerabilities such as adversarial attacks, model inversions and LLM poisoning. These tools include differential privacy and secure multi-party computation that are integrated into AI systems for training and deployment data protection purposes.
Request Sample Pages@ https://www.marketsandmarkets.com/requestsampleNew.asp?id=164202814
Generative AI apps security segment will account for largest market share during the forecast period.
The cybersecurity landscape is rapidly changing for generative AI apps, which are already making their way into chatbots, content creation tools like word processors, and personalized recommendation systems. According to McAfee, 55% of these programs have had security breaches. This highlights the dire need for stronger protective measures from unauthorized access. Several generative AI applications that use adversarial techniques to force the desired reaction out of intelligent machines.
Therefore, there is a pressing demand in the number of developers who ensure that such machines are made more robust through techniques like adversarially trained models and resistant architectures. Finally, the usage of secure enclaves plus hardware-based security measures is growing off late, mainly aimed at safeguarding vulnerable AI computations from being tampered with. For instance, OpenAI has very strict security rules meant to protect GPT models thereby ensuring data integrity and user privacy.
By end-user, government & defense sector is poised to account for larger market share in 2024.
Government as well as defense industries are increasingly resorting to generative AI for cyber security purposes due to the urgency of protecting sensitive information and national security. According to a recent CSIS report, AI is being integrated into the cybersecurity framework of 43% of government agencies which resultantly improves their ability to identify and counter threats. As an example, the United States Department of Defense has started using artificial intelligence (AI) based security solutions backed by generative AI that can create fictitious cyber-attacks, thereby providing them with enhanced preparedness against advanced types of threats.
This technology also helps these sectors handle and analyze large volumes of data more effectively, giving valuable insights that will enable them prevent or mitigate cyber threats. This trend demonstrates an increasing reliance on generative AI in fortifying cyber security measures so as to ensure that critical infrastructure and sensitive data remain secure in today’s intricate digital landscape.
By region, North America to hold the largest share by market value in 2024.
In 2024, North America will be the leading region based on market share due to its excellent technology infrastructure, substantial investments in AI-enabled cybersecurity and the presence of key players. Major cyber security research universities and tech companies such as Google, AWS, CrowdStrike, SentinelOne and IBM are present in this area, pushing them on the forefront of potent risk management technologies and generative AI tools for threat detection. For example, IBM’s security platform powered by AI has improved detection rates for threats up by 40%, thus proving the relevance of AI technology to enhancing cybersecurity.
Moreover, legislative instruments such as Cybersecurity Information Sharing Act (CISA) are being put in place to promote advanced cybersecurity technologies. As internet attacks continue getting more complicated, North American enterprises prefer generative artificial intelligence (AI), so as to enhance their safety measures pertaining to personal data and digital infrastructure.
Inquire Before Buying@ https://www.marketsandmarkets.com/Enquiry_Before_BuyingNew.asp?id=164202814
Top Key Companies in Generative AI cybersecurity Market:
The major players in the generative AI cybersecurity market include Palo Alto Networks (US), AWS (US), CrowdStrike (US), SentinelOne (US), and Google (US), along with SMEs and startups such as MOSTLY AI (Austria), XenonStack (UAE), BigID (US), Abnormal Security (US), and Adversa AI (Israel).
Browse Adjacent Market: Artificial Intelligence (AI) Market Research Reports & Consulting
Browse Other Reports:
AI Model Risk Management Market – Global Forecast to 2029
AI in Chemicals Market – Global Forecast to 2029
Artificial Intelligence in Cybersecurity Market – Global Forecast to 2028
Explainable AI Market – Global Forecast to 2028
Artificial Intelligence (AI) Toolkit Market – Global Forecast to 2028
Get access to the latest updates on Generative AI cybersecurity Companies and Generative AI cybersecurity Industry
About MarketsandMarkets™
MarketsandMarkets™ has been recognized as one of America’s best management consulting firms by Forbes, as per their recent report.
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. We have the widest lens on emerging technologies, making us proficient in co-creating supernormal growth for clients.
Earlier this year, we made a formal transformation into one of America’s best management consulting firms as per a survey conducted by Forbes.
The B2B economy is witnessing the emergence of $25 trillion of new revenue streams that are substituting existing revenue streams in this decade alone. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines – TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the ‘GIVE Growth’ principle, we work with several Forbes Global 2000 B2B companies – helping them stay relevant in a disruptive ecosystem. Our insights and strategies are molded by our industry experts, cutting-edge AI-powered Market Intelligence Cloud, and years of research. The KnowledgeStore™ (our Market Intelligence Cloud) integrates our research, facilitates an analysis of interconnections through a set of applications, helping clients look at the entire ecosystem and understand the revenue shifts happening in their industry.
To find out more, visit www.MarketsandMarkets™.com or follow us on Twitter, LinkedIn and Facebook.
Contact:Mr. Rohan SalgarkarMarketsandMarkets™ INC.630 Dundee RoadSuite 430Northbrook, IL 60062USA: +1-888-600-6441Email: [email protected] Our Website: https://www.marketsandmarkets.com/
Logo: https://mma.prnewswire.com/media/1951202/4609423/MarketsandMarkets.jpg
View original content:https://www.prnewswire.co.uk/news-releases/generative-ai-cybersecurity-market-worth-40-1-billion-by-2030—exclusive-report-by-marketsandmarkets-302207361.html
-

 Artificial Intelligence3 days ago
Artificial Intelligence3 days agoaelf Welcomes First COO to Foster AI and Blockchain Synergies
-

 Artificial Intelligence3 days ago
Artificial Intelligence3 days agoSINGAPORE INKS MOU WITH QUANTINUUM, ENABLING ACCESS TO THEIR ADVANCED QUANTUM COMPUTER
-

 Artificial Intelligence4 days ago
Artificial Intelligence4 days agoCandyspace strengthens ecommerce capabilities with the acquisition of Profound
-

 Artificial Intelligence4 days ago
Artificial Intelligence4 days agoEmmes Group partners with Miimansa AI to accelerate adoption of Generative AI in clinical research
-

 Artificial Intelligence4 days ago
Artificial Intelligence4 days agoDeep North Rebrands as StrataVision
-

 Artificial Intelligence5 days ago
Artificial Intelligence5 days agoWego Announces Partnership with Visit Valencia to Promote the Spanish City as a Premier Travel Destination
-

 Uncategorized1 day ago
Uncategorized1 day agoHow Hong Kong’s AI Data Guidelines Can Help Firms Embrace the Future
-

 Artificial Intelligence4 days ago
Artificial Intelligence4 days agoGreenRoad Ushers in New Era in Fleet Safety with Digital Edge™























